
Product data
| Category: | Respiratory Combo Test |
| Manufacturer: | Dvot |
| SKU: | FC-MAT01 |
| EAN: | 6973083141219 |
| Best before: | 30.04.2027 |
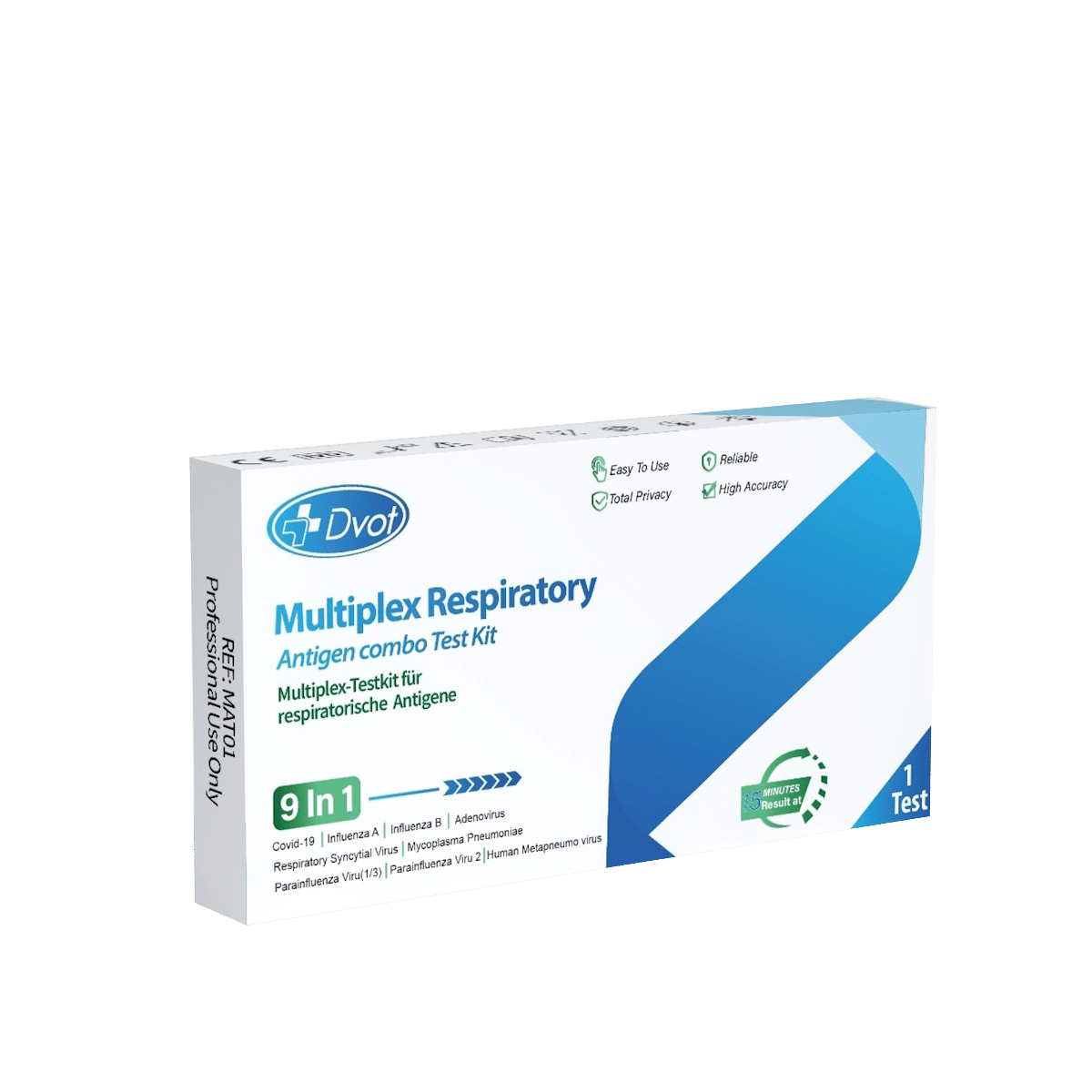
Dvot Multiplex Test Kit for Respiratory Antigens 9in1 (Professional Use)
Benefits:
- Multi-pathogen detection (9 tests in 1)
- Rapid results with lateral flow immunoassay
- CE certified for professional IVD use
- Ready-to-use kit with pre-filled extraction buffer
- Nasal and nasopharyngeal swab compatible
- No laboratory required
Tax included. Tax excluded.Shipping calculated at checkout
Volume Discounts
Volume Discounts
Shop safely
Volume Discounts
Volume Discounts
Description
The Dvot Multiplex Test Kit for Respiratory Antigens 9in1 is a comprehensive lateral flow immunoassay for the simultaneous qualitative detection of nine major respiratory pathogens in a single test cassette. Designed for professional use in medical practices, clinics, emergency departments, and laboratories, this CE-certified point-of-care diagnostic tool enables rapid differential diagnosis of respiratory infections without requiring laboratory equipment. Buy the Dvot 9in1 multiplex test today for reliable, fast, and resource-efficient respiratory pathogen screening.
Highlights & Benefits
- 9-in-1 Multi-Pathogen Detection: Simultaneously identifies SARS-CoV-2, Influenza A, Influenza B, RSV, Parainfluenza 1–3, Human Metapneumovirus (hMPV), Adenovirus, and Mycoplasma pneumoniae from a single sample.
- Rapid Results: Lateral flow immunoassay with clear line indication delivers actionable results within the specified incubation time—no waiting for lab processing.
- Ready-to-Use Kit: Pre-filled extraction buffer, sterile swabs, and test cassettes included. No additional reagents or equipment required.
- Flexible Sample Types: Compatible with both nasal and nasopharyngeal swabs, allowing clinicians to choose the most appropriate collection method.
- Cost & Time Efficient: One multiplex test replaces up to nine individual tests, significantly reducing costs, time, and consumable waste.
- Built-In Quality Control: Integrated control line confirms sufficient sample volume and validates correct test procedure for every test run.
- CE Certified: Meets European regulatory standards for in vitro diagnostic medical devices.
Who Should Use the Dvot 9in1 Test?
- General Practitioners & Urgent Care: Differential diagnosis of acute respiratory symptoms during cold, flu, and RSV season without sending samples to external labs.
- Emergency Departments: Rapid triage of respiratory infections to prioritize isolation measures and further diagnostic workup.
- Pediatric Clinics: Broad coverage of common childhood respiratory pathogens including RSV, Adenovirus, Parainfluenza, and Mycoplasma pneumoniae.
- Care Facilities & Nursing Homes: Outbreak investigation and cluster analysis when multiple pathogens may be circulating simultaneously.
- Occupational Health Services: Rapid point-of-care screening to support return-to-work decisions and infection control protocols.
- Laboratories & Reference Centers: First-line screening tool before confirmatory PCR testing.
Detected Pathogens
The Dvot 9in1 Multiplex Test covers the most clinically relevant respiratory pathogens in a single cassette:
- SARS-CoV-2 (COVID-19)
- Influenza A
- Influenza B
- RSV (Respiratory Syncytial Virus)
- Parainfluenza Virus Type 1
- Parainfluenza Virus Type 2
- Parainfluenza Virus Type 3
- Human Metapneumovirus (hMPV)
- Adenovirus
- Mycoplasma pneumoniae
How to Use
Test procedure follows the manufacturer's instructions for professional use:
Step 1: Sample Collection
Collect a nasal or nasopharyngeal swab using the sterile swab provided in the kit. Follow standard clinical swabbing protocols for optimal sample quality.
Step 2: Sample Extraction
Insert the swab into the pre-filled extraction buffer tube and rotate vigorously to ensure thorough sample release. Remove and discard the swab according to biosafety guidelines.
Step 3: Apply to Cassette
Dispense the required number of drops from the extraction tube into the sample well of the test cassette.
Step 4: Read Results
After the specified incubation time, interpret results by reading test and control lines. The control line (C) must appear for a valid test. Any visible test line indicates a positive result for the corresponding pathogen.
Package Contents
- 1 × Dvot 9in1 Multiplex Test Cassette (individually sealed)
- 1 × Sterile Swab
- 1 × Pre-filled Extraction Buffer Tube
- 1 × Instructions for Use (IFU)
Available Variants
- Dvot Multiplex Test Kit for Respiratory Antigens 9in1 (Professional Use) – single test
Notes
- For in vitro diagnostic purposes and professional use only
- Store according to manufacturer specifications (2–30°C)
- Do not use if packaging is damaged or the test has expired
- Test results should be evaluated in clinical context; confirm with PCR or other laboratory methods if needed
- Dispose of used test materials according to local biosafety regulations
- This product complies with European in vitro diagnostic regulations
Product data
| Category: | Respiratory Combo Test |
| Manufacturer: | Dvot |
| SKU: | FC-MAT01 |
| EAN: | 6973083141219 |
| Best before: | 30.04.2027 |
Regulatory notices
- CE-certified in-vitro diagnostic (IVD)
- Intended for self-testing by lay users
- For single use only
- Read instructions carefully before use
- Store at room temperature (2-30 degC)
- Please note that the product you have selected is subject to a limitation of the right of return according to Sec. 312g para. 2 no. 3 BGB. As this product is a medical device, returns are excluded for hygiene reasons.
Sales inquiry / B2B
Interested in bulk orders or reseller pricing? Contact us – we will get back to you as soon as possible.
Or email us directly at sales@parahealth.de
Frequently asked questions about this product
What is the Dvot 9in1 Multiplex Respiratory Test?
The Dvot 9in1 Multiplex Test is a CE-certified lateral flow immunoassay that simultaneously detects nine respiratory pathogens from a single nasal or nasopharyngeal swab: SARS-CoV-2 (COVID-19), Influenza A, Influenza B, RSV, Parainfluenza 1-3, Human Metapneumovirus (hMPV), Adenovirus, and Mycoplasma pneumoniae. Designed exclusively for professional use in clinics, practices, and laboratories.
How does the Dvot multiplex test work?
Collect a nasal or nasopharyngeal swab using the sterile swab provided. Insert the swab into the pre-filled extraction buffer and rotate vigorously to release the sample. Apply the recommended number of drops to the sample well on the test cassette. After the specified incubation time, read the results: the control line (C) must appear for a valid test, and any visible test line indicates a positive result for that specific pathogen.
Which pathogens does the Dvot 9in1 test detect?
The test covers nine clinically relevant respiratory pathogens: SARS-CoV-2 (COVID-19), Influenza A, Influenza B, RSV (Respiratory Syncytial Virus), Parainfluenza Virus Types 1, 2, and 3, Human Metapneumovirus (hMPV), Adenovirus, and Mycoplasma pneumoniae. This broad panel enables comprehensive differential diagnosis of acute respiratory infections.
Where can I buy the Dvot 9in1 test?
You can purchase the Dvot 9in1 Multiplex Respiratory Test directly at parahealth.de. We offer volume discounts for larger orders and free shipping on orders over 100 EUR. Orders are typically dispatched within 1-2 business days from our Berlin warehouse.
Who can use this test?
The Dvot 9in1 Multiplex Test is designed exclusively for professional use by trained healthcare personnel. It is intended for use in medical practices, clinics, emergency departments, laboratories, nursing homes, and occupational health services. This test is not intended for self-testing or home use.
What is included in the Dvot test kit?
Each Dvot 9in1 test kit contains: 1 individually sealed multiplex test cassette with result windows for all nine pathogens, 1 sterile swab for sample collection, 1 pre-filled extraction buffer tube, and 1 detailed instructions for use (IFU). No additional reagents or laboratory equipment are required.
Frequently Asked Questions
Find answers to the most common questions about ordering, shipping, and our products.
Are your products clinically tested and safe?
Yes, all our medical devices and diagnostic tests undergo rigorous clinical testing and validation to ensure safety, accuracy, and efficacy. We comply with all relevant medical device regulations and quality standards.
What is your return policy?
We offer a 14-day return policy for private customers. Products must be unused and in original packaging. Please note that B2B and wholesale customers are subject to different terms. Contact our wholesale team for details.
How long does shipping take to Germany?
Standard shipping to Germany takes 1-2 business days from our Berlin facility. We also offer expedited options at checkout for faster delivery.
Do you provide support for professional/clinical use?
Yes, we offer dedicated support for healthcare professionals and clinics, including product training and technical assistance.
What are your B2B payment and shipping terms?
B2B customers receive customized payment terms and shipping arrangements. Please contact our wholesale team for details.
Do you offer bulk orders or wholesale pricing?
Yes, we offer B2B wholesale pricing for pharmacies, clinics, and medical practices. Contact us for volume pricing and terms.
Will I receive tracking information?
Yes, all orders include tracking information sent via email so you can monitor your shipment.
Do you ship internationally?
Yes, we ship worldwide from our Berlin facility. International shipping times vary by destination.
Where do you ship from?
We ship from our state-of-the-art AutoStore Warehouse in Berlin, Germany, ensuring fast and efficient order processing and delivery.
